NPs Basic Information
|
Name |
Exophillic acid
|
| Molecular Formula | C38H56O12 | |
| IUPAC Name* |
2-hydroxy-4-[4-hydroxy-2-nonyl-6-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxybenzoyl]oxy-6-nonylbenzoic acid
|
|
| SMILES |
CCCCCCCCCC1=C(C(=CC(=C1)O)O[C@H]2[C@@H]([C@H]([C@@H]([C@H](O2)CO)O)O)O)C(=O)OC3=CC(=C(C(=C3)O)C(=O)O)CCCCCCCCC
|
|
| InChI |
InChI=1S/C38H56O12/c1-3-5-7-9-11-13-15-17-24-19-26(40)21-29(49-38-35(44)34(43)33(42)30(23-39)50-38)32(24)37(47)48-27-20-25(31(36(45)46)28(41)22-27)18-16-14-12-10-8-6-4-2/h19-22,30,33-35,38-44H,3-18,23H2,1-2H3,(H,45,46)/t30-,33-,34+,35-,38-/m1/s1
|
|
| InChIKey |
GBXMNTLQBMLGFM-IHQQTPIISA-N
|
|
| Synonyms |
Exophillic acid; 2-hydroxy-4-[4-hydroxy-2-nonyl-6-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxybenzoyl]oxy-6-nonylbenzoic acid; 2-hydroxy-4-[4-hydroxy-2-nonyl-6-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydropyran-2-yl]oxy-benzoyl]oxy-6-nonyl-benzoic acid; Benzoic acid, 2-(.beta.-D-glucopyranosyloxy)-4-hydroxy-6-nonyl-, 4-carboxy-3-hydroxy-5-nonylphenyl ester
|
|
| CAS | NA | |
| PubChem CID | 3008194 | |
| ChEMBL ID | NA |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 704.8 | ALogp: | 9.7 |
| HBD: | 7 | HBA: | 12 |
| Rotatable Bonds: | 23 | Lipinski's rule of five: | Rejected |
| Polar Surface Area: | 203.0 | Aromatic Rings: | 3 |
| Heavy Atoms: | 50 | QED Weighted: | 0.044 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -5.863 | MDCK Permeability: | 0.00001290 |
| Pgp-inhibitor: | 0.896 | Pgp-substrate: | 0.941 |
| Human Intestinal Absorption (HIA): | 0.922 | 20% Bioavailability (F20%): | 0.999 |
| 30% Bioavailability (F30%): | 0.998 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.022 | Plasma Protein Binding (PPB): | 99.99% |
| Volume Distribution (VD): | 0.364 | Fu: | 0.67% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.247 | CYP1A2-substrate: | 0.083 |
| CYP2C19-inhibitor: | 0.073 | CYP2C19-substrate: | 0.055 |
| CYP2C9-inhibitor: | 0.191 | CYP2C9-substrate: | 0.332 |
| CYP2D6-inhibitor: | 0.873 | CYP2D6-substrate: | 0.11 |
| CYP3A4-inhibitor: | 0.084 | CYP3A4-substrate: | 0.002 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 1.826 | Half-life (T1/2): | 0.593 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.49 | Human Hepatotoxicity (H-HT): | 0.193 |
| Drug-inuced Liver Injury (DILI): | 0.966 | AMES Toxicity: | 0.092 |
| Rat Oral Acute Toxicity: | 0.01 | Maximum Recommended Daily Dose: | 0.035 |
| Skin Sensitization: | 0.899 | Carcinogencity: | 0.023 |
| Eye Corrosion: | 0.003 | Eye Irritation: | 0.054 |
| Respiratory Toxicity: | 0.086 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
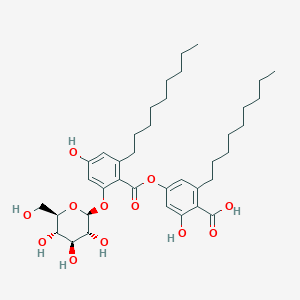
| ENC003312 | 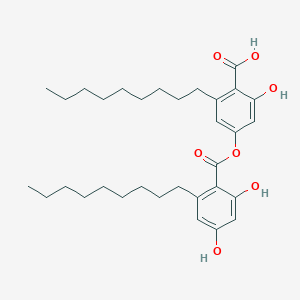 |
0.722 | D00STJ |  |
0.375 | ||
| ENC003334 |  |
0.448 | D0T9TJ |  |
0.302 | ||
| ENC002894 | 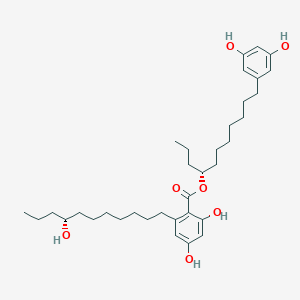 |
0.444 | D07ILQ |  |
0.301 | ||
| ENC004641 | 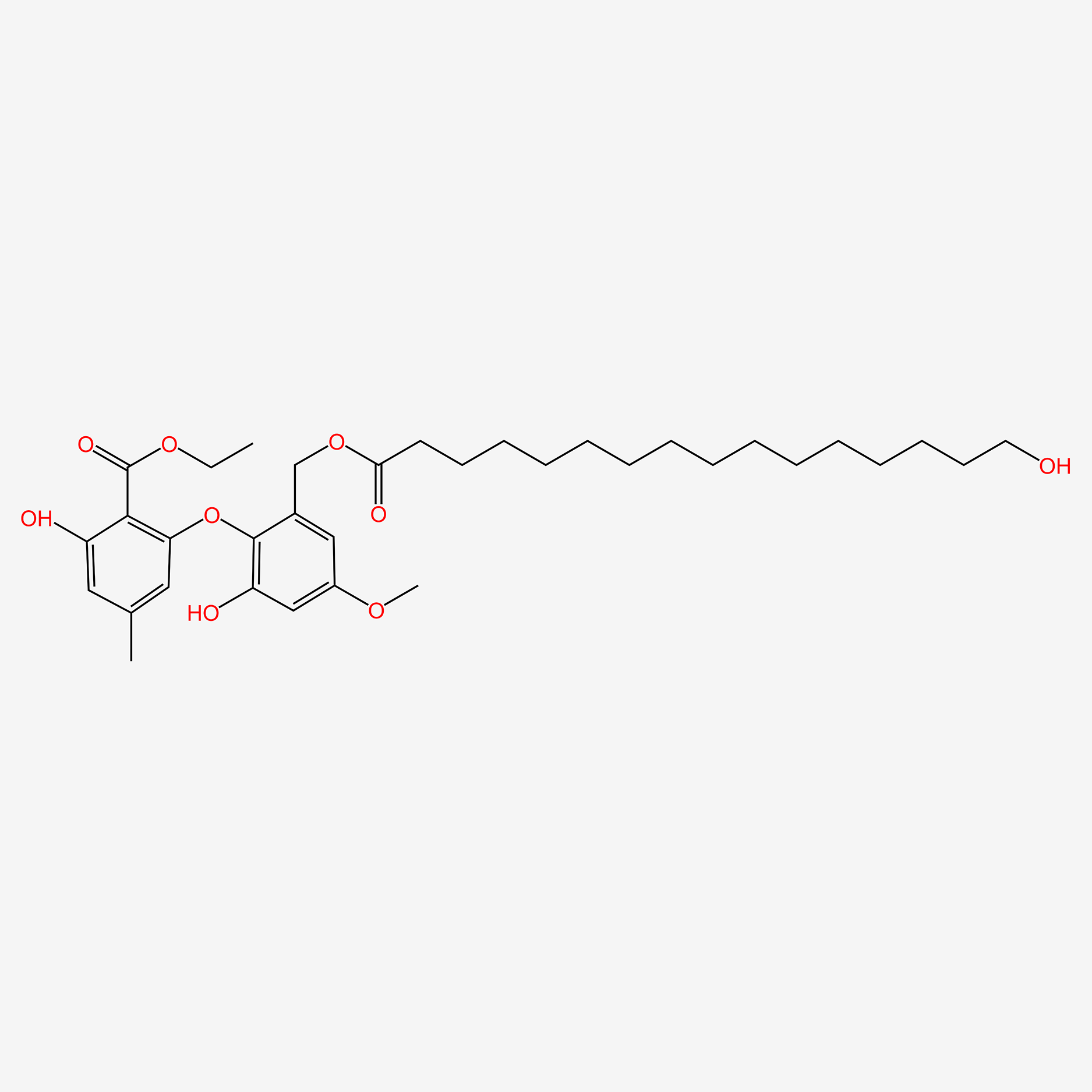 |
0.436 | D06BQU | 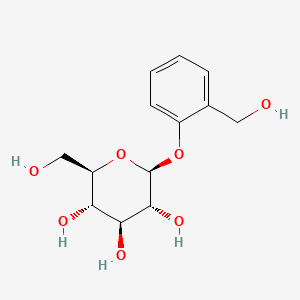 |
0.276 | ||
| ENC005852 | 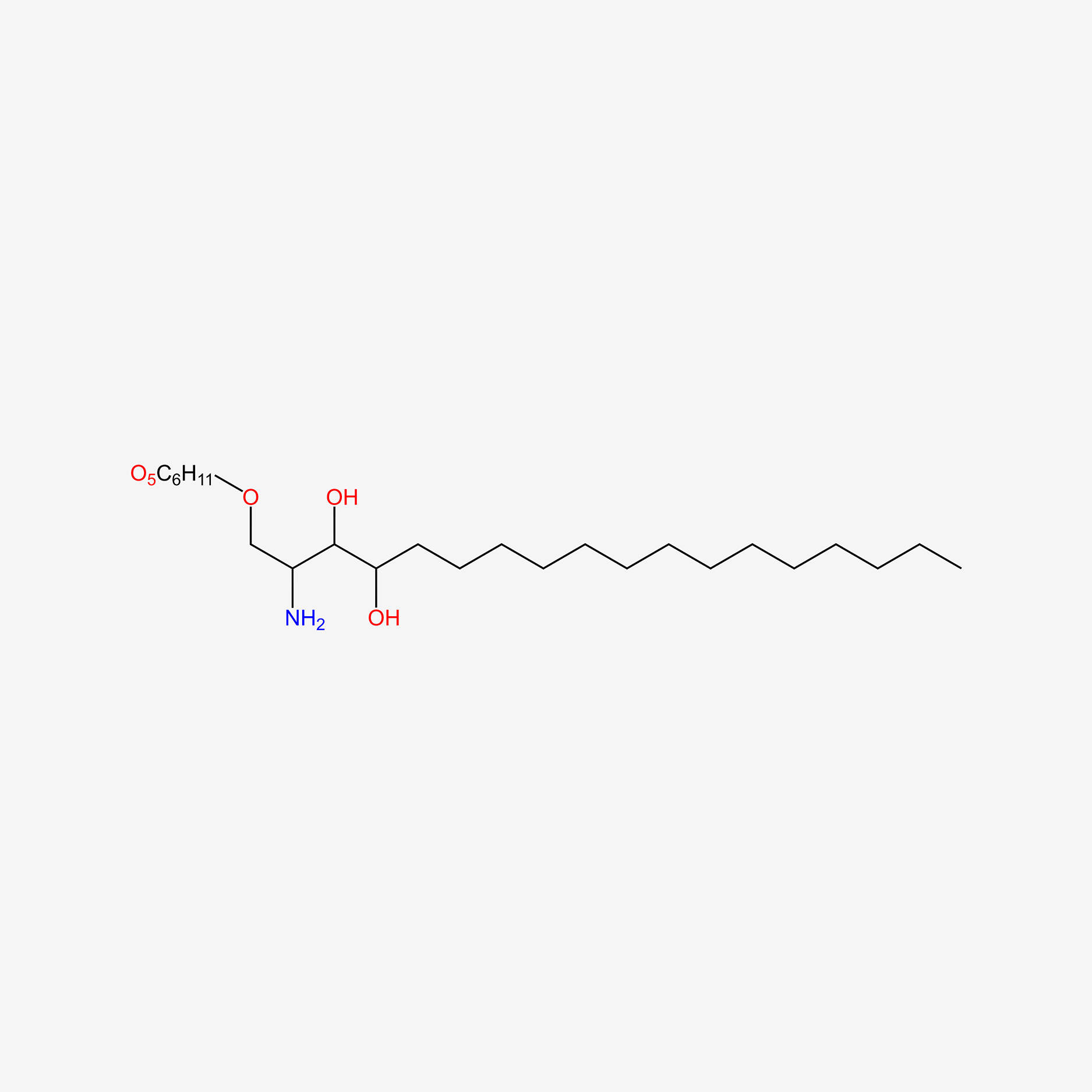 |
0.429 | D00FGR |  |
0.274 | ||
| ENC004642 | 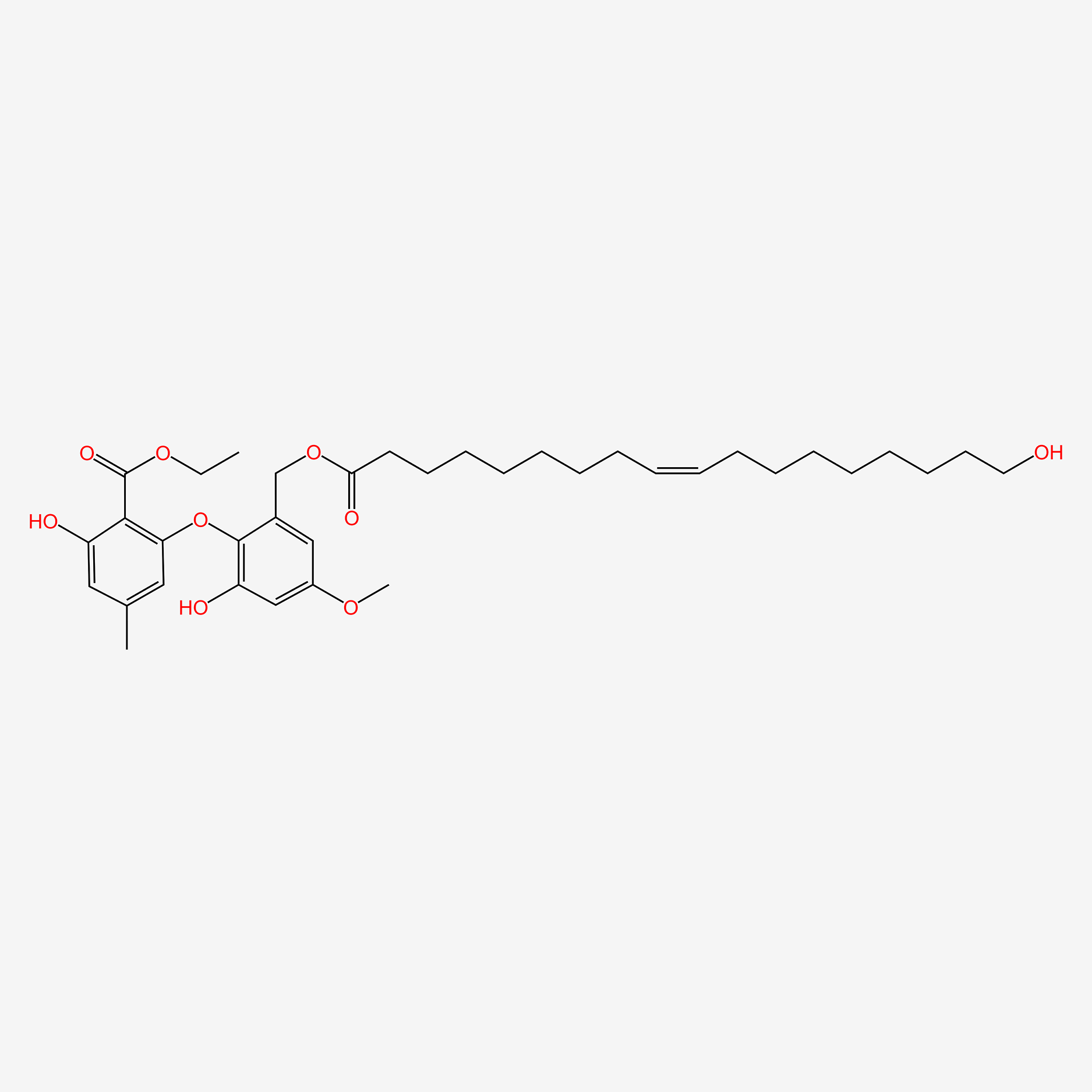 |
0.407 | D0O1PH |  |
0.273 | ||
| ENC002672 | 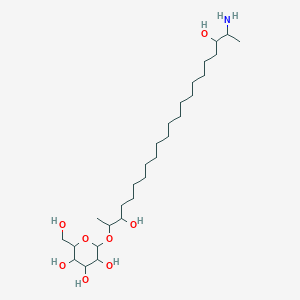 |
0.401 | D0P1RL |  |
0.273 | ||
| ENC002909 |  |
0.391 | D0TC7C |  |
0.273 | ||
| ENC002194 |  |
0.391 | D0I9HF |  |
0.272 | ||
| ENC005817 | 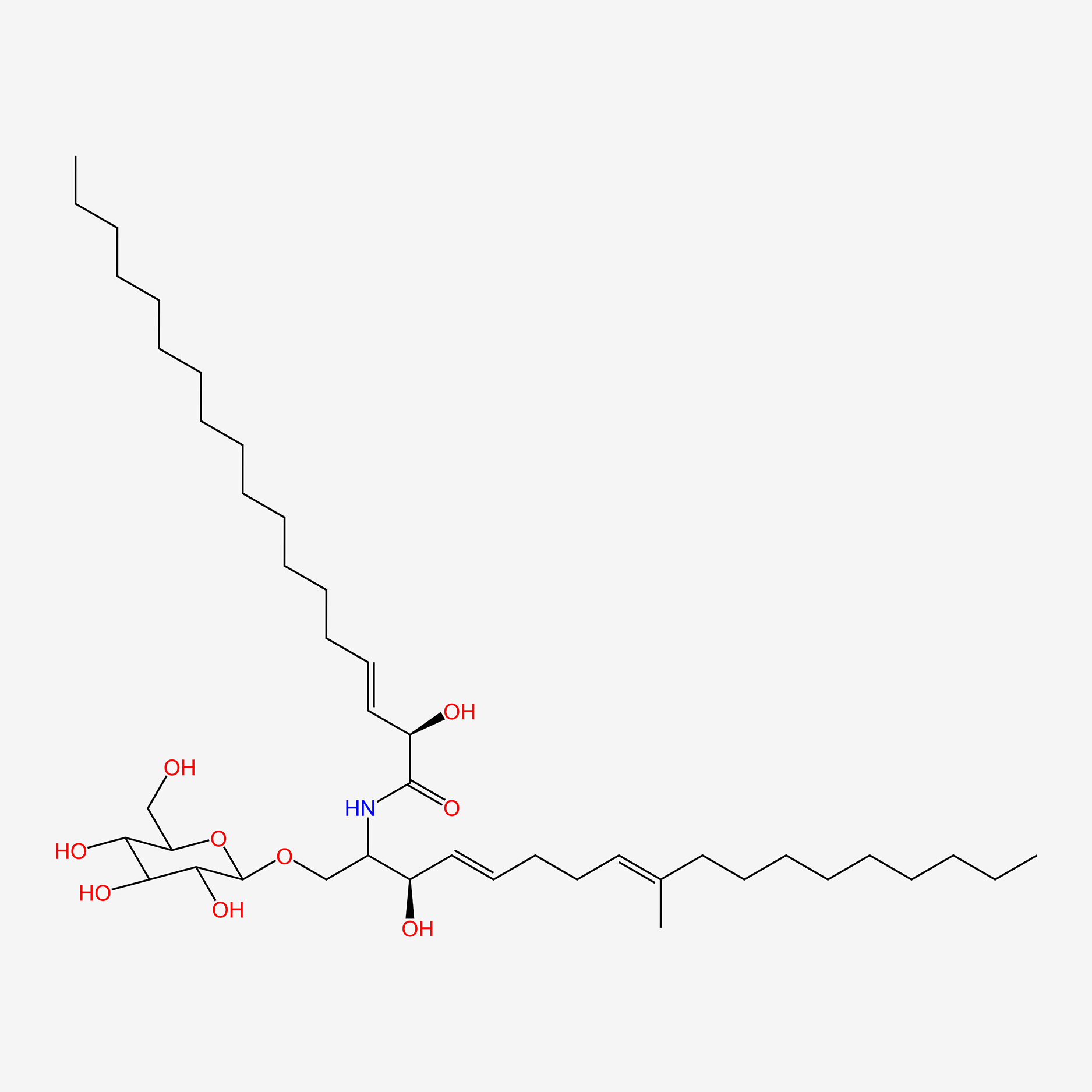 |
0.391 | D00AOJ |  |
0.272 | ||