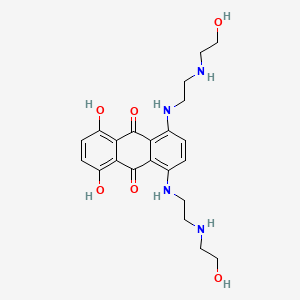
mitoxantrone, 65271-80-9, Mitozantrone, Mitoxanthrone, Mitoxantron, DHAQ, Mitoxantrona, Mitoxantronum, Misostol, Mitoxantrone [INN], Mitoxantronum [INN-Latin], Mitoxantrona [INN-Spanish], CCRIS 7604, NSC 279836, NSC-279836, UNII-BZ114NVM5P, DHAD, BZ114NVM5P, NSC279836, 1,4-Bis(2-(2-hydroxyethylamino)ethyl)amino)-5,8-dihydroxyanthraquinone, 1,4-Dihydroxy-5,8-bis[2-(2-hydroxyethylamino)ethylamino]anthracene-9,10-dione, BRN 2795126, Mitoxantrone (INN), Mitoxantrone free base, CHEBI:50729, 5,8-Bis((2-((2-hydroxyethyl)amino)ethyl)amino)-1,4-dihydroxyanthraquinone, 1,4-Dihydroxy-5,8-bis(2-((2-hydroxyethyl)amino)ethylamino)-9,10-anthracenedione, 1,4-Dihydroxy-5,8-bis(5-hydroxy-3-azapentylamino)anthrachinon, 1,4-dihydroxy-5,8-bis((2-((2-hydroxyethyl)amino)ethyl)amino)anthracene-9,10-dione, 1,4-DIHYDROXY-5,8-BIS({2-[(2-HYDROXYETHYL)AMINO]ETHYL}AMINO)-9,10-ANTHRACENEDIONE, 9,10-Anthracenedione, 1,4-dihydroxy-5,8-bis((2-((2-hydroxyethyl)amino)ethyl)amino)-, MLS002703044, DTXSID4046947, 1,4-dihydroxy-5,8-bis[2-[(1,1,2,2-tetradeuterio-2-hydroxyethyl)amino]ethylamino]anthracene-9,10-dione, 65271-80-9 (free base), MFCD00242942, 1,4-dihydroxy-5,8-bis({2-[(2-hydroxyethyl)amino]ethyl}amino)anthracene-9,10-dione, ANTHRAQUINONE, 5,8-BIS((2-((2-HYDROXYETHYL)AMINO)ETHYL)AMINO)-1,4-DIHYDROXY-, NSC299195, NSC301739, MITOXANTRONE (IARC), MITOXANTRONE [IARC], Mitoxantronum (INN-Latin), Mitoxantrona (INN-Spanish), 1,4-Dihydroxy-5,8-bis[[2-[(2-hydroxyethyl)amino]ethyl]amino]-9,10-anthracenedione, 1,4-dihydroxy-5,8-bis({2-[(2-hydroxyethyl)amino]ethyl}amino)-9,10-dihydroanthracene-9,10-dione, MIX, MLS001333711, 9,10-Anthracenedione, 1,4-dihydroxy-5,8-bis[[2-[(2-hydroxyethyl)amino]ethyl]amino]-, Misostol (TN), NCGC00015693-02, Mitoxantrone (free base), SMR000058480, Mitoxantrone [INN:BAN], CAS-70476-82-3, DHAQ (*Diacetate salt*), SR-01000076001, Mitoxantrone base, 2fum, 1,4-DIHYDROXY-5,8-BIS((2-((2-HYDROXYETHYL)AMINO)ETHYL)AMINO)-9,10-ANTHRACENEDIONE, 1,4-dihydroxy-5,8-bis(2-(2-hydroxyethylamino)ethylamino)anthracene-9,10-dione, 1,4-Dihydroxy-5,8-bis[[2-[(2-hydroxyethyl)amino]ethyl]amino]anthracene-9,10-dione, Immunex (Salt/Mix), dihydroxyanthracenedione, Spectrum_001655, CHEMBL58, Prestwick0_000385, Prestwick1_000385, Prestwick2_000385, Prestwick3_000385, Spectrum2_000908, Spectrum3_001590, Spectrum4_000866, Spectrum5_001205, Lopac-M-6545, MITOXANTRONE [MI], cid_4212, Neuro_000153, SCHEMBL3000, MITOXANTRONE [VANDF], BIDD:PXR0181, Lopac0_000779, BSPBio_000569, BSPBio_003160, KBioGR_001531, KBioSS_002135, DivK1c_000516, MITOXANTRONE [WHO-DD], SPBio_000756, SPBio_002490, BPBio1_000627, GTPL7242, DTXCID2026947, BDBM67690, cid_5458171, KBio1_000516, KBio2_002135, KBio2_004703, KBio2_007271, KBio3_002660, L01DB07, NINDS_000516, GLXC-03038, HMS2090D05, HMS3655E20, KUC108634N, BCP02861, CCG-36371, s1889, STK631833, Mitoxantrone hydrochloride (Salt/Mix), AKOS005564036, BCP9000931, DB01204, KSC-19-204, SDCCGSBI-0050757.P004, IDI1_000516, SMP2_000179, NCGC00015693-01, NCGC00015693-03, NCGC00015693-04, NCGC00015693-05, NCGC00015693-06, NCGC00015693-08, NCGC00015693-13, NCGC00015693-23, NCGC00162251-01, AS-35321, HY-13502, MITOXANTRONE, Mitoxantrone Hydrochloride, Mitoxantrone dihydrochloride, MITOXANTHRONE HYDROCHLORIDE, NCI60_002276, NCI60_002535, SMR001549953, SY226320, SBI-0050757.P003, AB00053716, FT-0630752, FT-0672423, M3133, NS00008439, SW196745-6, VU0244399-2, D08224, EN300-117257, AB00053716-18, AB00053716-19, AB00053716-22, AB00053716_23, AB00053716_24, Q239426, SR-01000076001-7, BRD-K21680192-001-01-5, BRD-K21680192-001-11-4, BRD-K21680192-300-05-2, BRD-K21680192-300-07-8, BRD-K21680192-300-09-4, BRD-K21680192-300-10-2, BRD-K21680192-300-12-8, 1,4-bis[2-(2-hydroxyethylamino)-ethylamino]-5,8-dihydroxyanthraquinone, 1,4-bis[2-(2-hydroxyethylamino)ethylamino]-5,8-dihydroxyanthraquinone, 1,4-dihydroxy-5,8-bis[[2-(2-hydroxyethylamino)ethyl]amino]anthraquinone, 9, 1,4-dihydroxy-5,8-bis[[2-[(2-hydroxyethyl)amino]ethyl] amino]-, 1,3-Dihydroxy-5,8-bis((2-((2-hydroxyethyl)amino)ethyl)amino)-9, 10-anthracenedione, 1,4-Dihydroxy-5,8-bis((2-((2-hydroxyethyl)amino)ethyl)amino)-9, 10-anthroquinone, 1,4-Dihydroxy-5,8-bis-[[2-[(2-hydroxyethyl)amino]ethyl]amino]anthraquinone, 1,4-dihydroxy-5,8-bis[2-(2-hydroxyethylamino)ethylamino]-9,10-anthraquinone;hydrochloride, 1,4-dihydroxy-5,8-bis[2-(2-hydroxyethylamino)ethylamino]anthracene-9,10-dione;hydrochloride, 1,4-bis[2-(2-hydroxyethylamino)ethylamino]-5,8-bis(oxidanyl)anthracene-9,10-dione;hydrochloride, MITOXANTRONE; 1,4-DIHYDROXY-5,8-BIS({2-[(2-HYDROXYETHYL)AMINO]ETHYL}AMINO)ANTHRA-9,10-QUINONE