NPs Basic Information
|
Name |
Benzydamine
|
| Molecular Formula | C19H23N3O | |
| IUPAC Name* |
3-(1-benzylindazol-3-yl)oxy-N,N-dimethylpropan-1-amine
|
|
| SMILES |
CN(C)CCCOC1=NN(C2=CC=CC=C21)CC3=CC=CC=C3
|
|
| InChI |
InChI=1S/C19H23N3O/c1-21(2)13-8-14-23-19-17-11-6-7-12-18(17)22(20-19)15-16-9-4-3-5-10-16/h3-7,9-12H,8,13-15H2,1-2H3
|
|
| InChIKey |
CNBGNNVCVSKAQZ-UHFFFAOYSA-N
|
|
| Synonyms |
BENZYDAMINE; 642-72-8; Benzidamine; Benzindamine; Benzydaminum; Bencidamina; Apo-Benzydamine; 3-(1-benzylindazol-3-yl)oxy-N,N-dimethylpropan-1-amine; 1-Benzyl-3-(3-(dimethylamino)propoxy)-1H-indazole; Benzydamine (INN); 1-Benzyl-3-[3-(dimethylamino)propoxy]-1H-indazole; 1-Propanamine, N,N-dimethyl-3-((1-(phenylmethyl)-1H-indazol-3-yl)oxy)-; 1H-Indazole, 1-benzyl-3-(3-(dimethylamino)propoxy)-; {3-[(1-benzyl-1H-indazol-3-yl)oxy]propyl}dimethylamine; 4O21U048EF; Benzidamina; Benzidamina [DCIT]; 3-[(1-benzyl-1H-indazol-3-yl)oxy]-N,N-dimethylpropan-1-amine; BENZYDAMINE [INN]; 1-Propanamine, N,N-dimethyl-3-[[1-(phenylmethyl)-1H-indazol-3-yl]oxy]-; Benzydamine [INN:BAN]; Benzydaminum [INN-Latin]; Bencidamina [INN-Spanish]; benzydamide; Apo-Benzydamine (TN); NCGC00016397-01; CAS-132-69-4; EINECS 211-388-8; C 1523; BRN 0896536; UNII-4O21U048EF; SR-01000799149; BENZYDAMINE [MI]; Prestwick0_000297; Prestwick1_000297; Prestwick2_000297; Prestwick3_000297; SCHEMBL26172; BENZYDAMINE [WHO-DD]; BSPBio_000234; 5-23-11-00248 (Beilstein Handbook Reference); CHEMBL12610; SPBio_002453; BPBio1_000258; DTXSID7047859; CHEBI:94563; HMS2090P22; ALBB-027259; ZINC2020083; BBL009932; BDBM50103598; STK711092; AKOS005530662; DB09084; SB16913; NCGC00016397-02; NCGC00016397-07; AC-15604; SBI-0206866.P001; FT-0740906; D07516; 1benzyl-3-[3-(dimethylamino)propoxy]-1H-indazole; 642B728; EN300-12576563; Q793143; SR-01000799149-2; BRD-K28542495-001-01-4; BRD-K28542495-003-03-6; BRD-K28542495-003-07-7; 1-BENZYL-1H-INDAZOL-3-YL 3-(DIMETHYLAMINO)PROPYL ETHER; 2-{[(2-Methoxyethyl)amino]-carbonyl}cyclohexanecarboxylicacid; 3-(1-benzyl-1H-indazol-3-yloxy)-N,N-dimethylpropan-1-amine; 3-[(1-benzyl-1H-indazol-3-yl)oxy]-N,N-dimethylpropan-1-aminie; N,N-dimethyl-3-[[1-(phenylmethyl)-3-indazolyl]oxy]-1-propanamine; N-(3-[(1-Benzyl-1H-indazol-3-yl)oxy]propyl)-N,N-dimethylamine #; N,N-DIMETHYL-3-((1-(PHENYLMETHYL)-1H-INDAZOL-3-YL)OXY)-1-PROPANAMINE
|
|
| CAS | 642-72-8 | |
| PubChem CID | 12555 | |
| ChEMBL ID | CHEMBL12610 |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 309.4 | ALogp: | 3.9 |
| HBD: | 0 | HBA: | 3 |
| Rotatable Bonds: | 7 | Lipinski's rule of five: | Accepted |
| Polar Surface Area: | 30.3 | Aromatic Rings: | 3 |
| Heavy Atoms: | 23 | QED Weighted: | 0.614 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.542 | MDCK Permeability: | 0.00002750 |
| Pgp-inhibitor: | 0.998 | Pgp-substrate: | 0.001 |
| Human Intestinal Absorption (HIA): | 0.003 | 20% Bioavailability (F20%): | 0.006 |
| 30% Bioavailability (F30%): | 0.003 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.991 | Plasma Protein Binding (PPB): | 92.70% |
| Volume Distribution (VD): | 3.422 | Fu: | 6.04% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.678 | CYP1A2-substrate: | 0.925 |
| CYP2C19-inhibitor: | 0.13 | CYP2C19-substrate: | 0.973 |
| CYP2C9-inhibitor: | 0.019 | CYP2C9-substrate: | 0.12 |
| CYP2D6-inhibitor: | 0.983 | CYP2D6-substrate: | 0.914 |
| CYP3A4-inhibitor: | 0.123 | CYP3A4-substrate: | 0.791 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 11.883 | Half-life (T1/2): | 0.042 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.953 | Human Hepatotoxicity (H-HT): | 0.074 |
| Drug-inuced Liver Injury (DILI): | 0.468 | AMES Toxicity: | 0.02 |
| Rat Oral Acute Toxicity: | 0.087 | Maximum Recommended Daily Dose: | 0.055 |
| Skin Sensitization: | 0.502 | Carcinogencity: | 0.103 |
| Eye Corrosion: | 0.003 | Eye Irritation: | 0.014 |
| Respiratory Toxicity: | 0.953 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
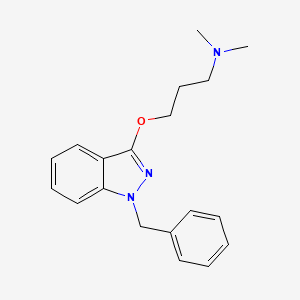
| ENC003208 | 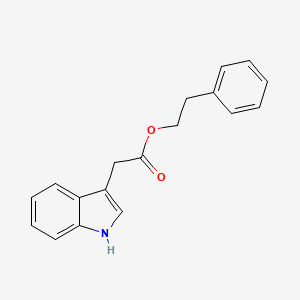 |
0.402 | D0KS6W | 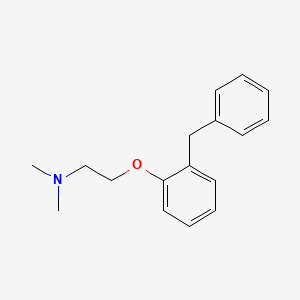 |
0.544 | ||
| ENC000302 |  |
0.384 | D0B4JQ |  |
0.452 | ||
| ENC000732 |  |
0.373 | D0D9FV | 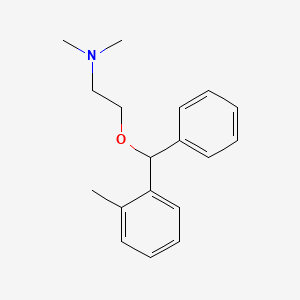 |
0.442 | ||
| ENC000908 | 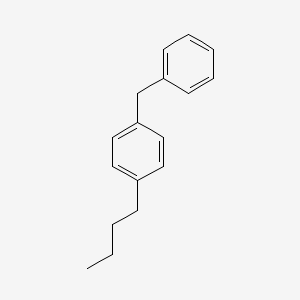 |
0.360 | D00NAX | 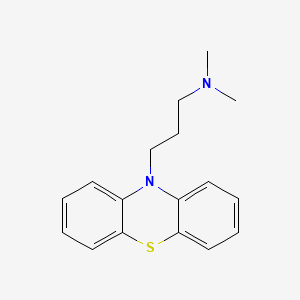 |
0.437 | ||
| ENC004267 |  |
0.358 | D01FGR | 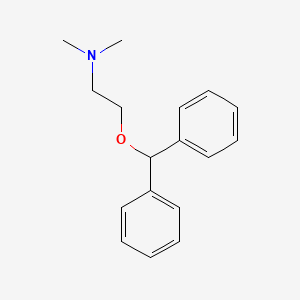 |
0.435 | ||
| ENC004606 | 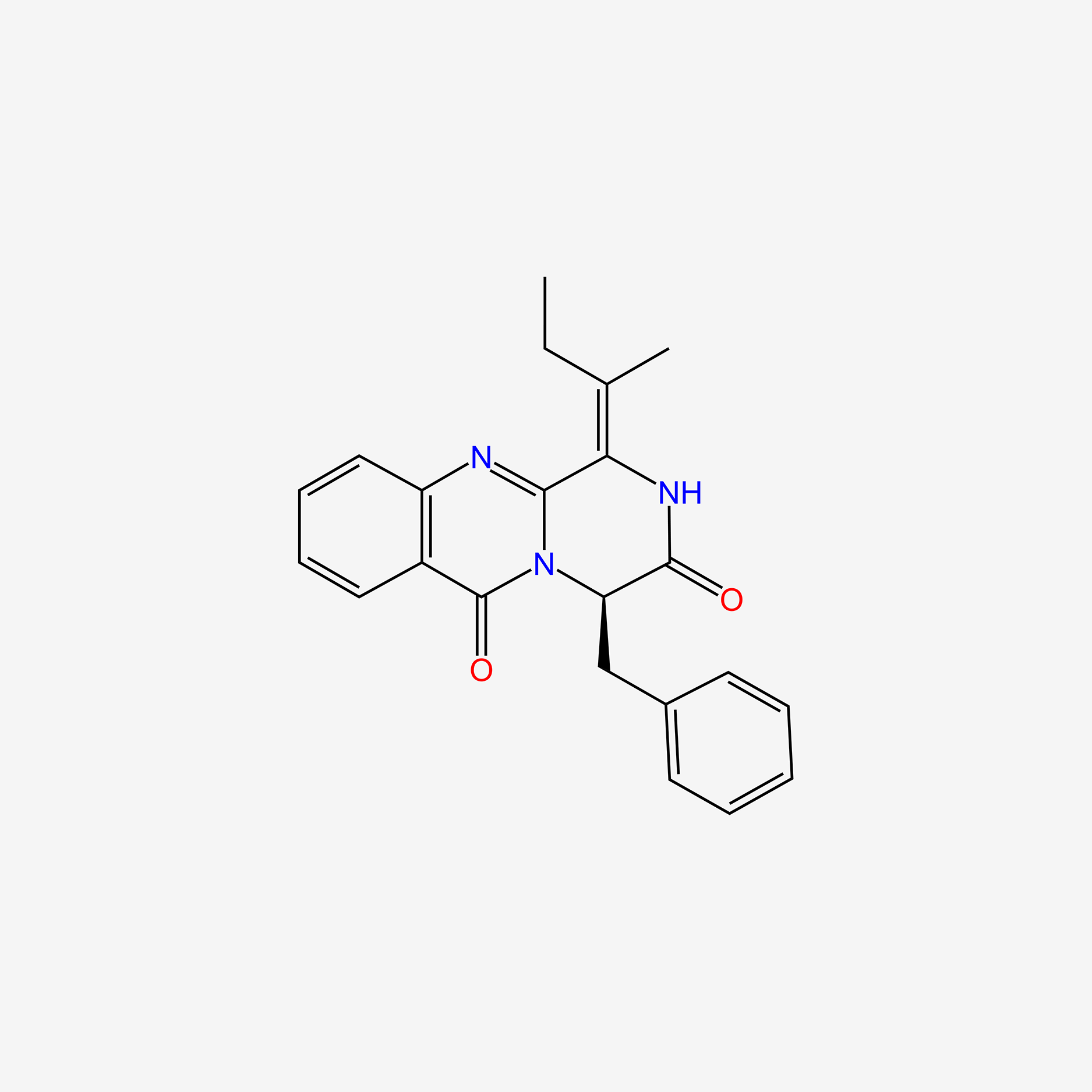 |
0.358 | D06ZUK | 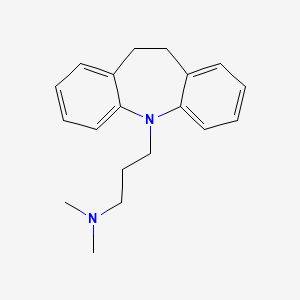 |
0.422 | ||
| ENC004605 | 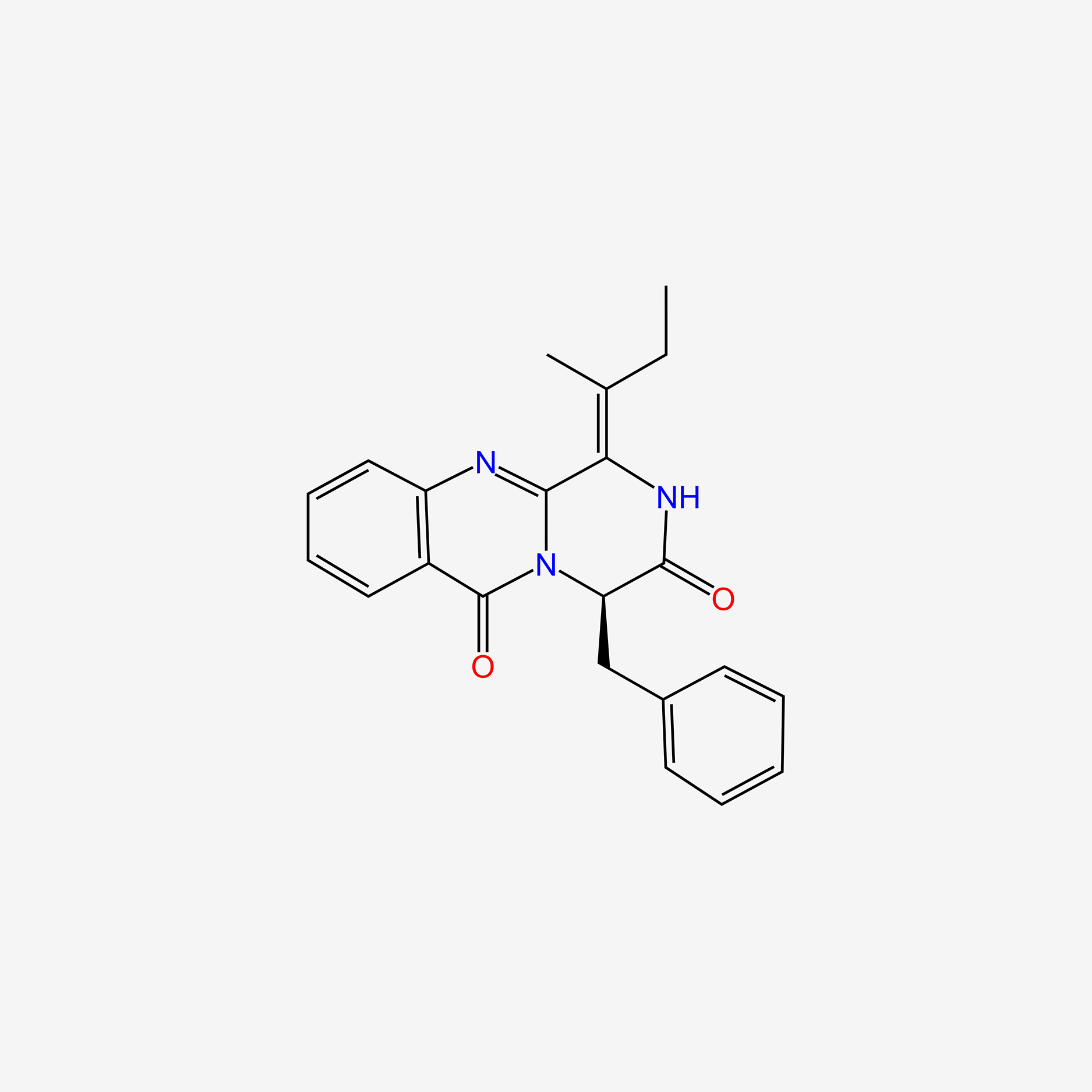 |
0.358 | D0EC6Q | 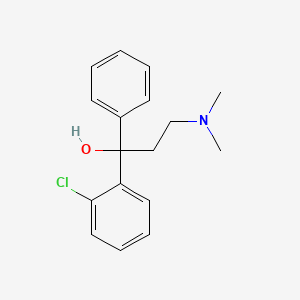 |
0.414 | ||
| ENC004646 | 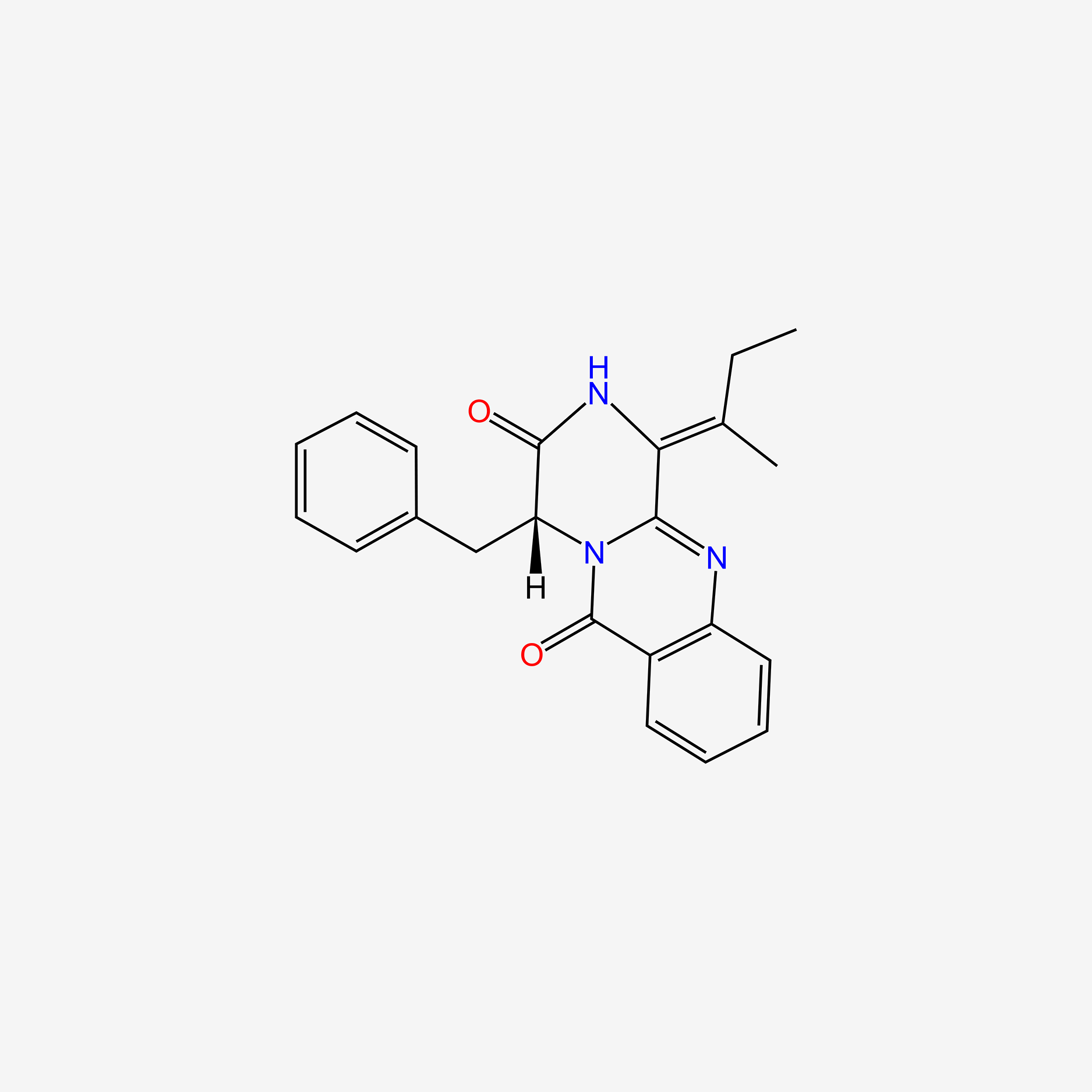 |
0.358 | D0H5LK |  |
0.411 | ||
| ENC000077 |  |
0.357 | D0Y2LR |  |
0.409 | ||
| ENC004348 |  |
0.355 | D0X2DK |  |
0.407 | ||