NPs Basic Information

|
Name |
Formamide, N-(1-methylpropyl)-
|
| Molecular Formula | C5H11NO | |
| IUPAC Name* |
N-butan-2-ylformamide
|
|
| SMILES |
CCC(C)NC=O
|
|
| InChI |
InChI=1S/C5H11NO/c1-3-5(2)6-4-7/h4-5H,3H2,1-2H3,(H,6,7)
|
|
| InChIKey |
NRMJMIGWXUCEEW-UHFFFAOYSA-N
|
|
| Synonyms |
53798-89-3; N-(butan-2-yl)formamide; Formamide, N-(1-methylpropyl)-; N-sec-Butylformamide; N-(2-Butyl)formamide; Formamide, N-sec-butyl-; NSC-404523; sec-butylformamide; NSC404523; H6SPH3V46T; SCHEMBL25180; DTXSID00323645; N-(1-METHYLPROPYL)FORMAMIDE; AKOS006274294; EN300-211462
|
|
| CAS | 53798-89-3 | |
| PubChem CID | 346326 | |
| ChEMBL ID | NA |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 101.15 | ALogp: | 0.9 |
| HBD: | 1 | HBA: | 1 |
| Rotatable Bonds: | 2 | Lipinski's rule of five: | Accepted |
| Polar Surface Area: | 29.1 | Aromatic Rings: | 0 |
| Heavy Atoms: | 7 | QED Weighted: | 0.526 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.327 | MDCK Permeability: | 0.00008380 |
| Pgp-inhibitor: | 0 | Pgp-substrate: | 0.382 |
| Human Intestinal Absorption (HIA): | 0.007 | 20% Bioavailability (F20%): | 0.001 |
| 30% Bioavailability (F30%): | 0.003 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.983 | Plasma Protein Binding (PPB): | 5.96% |
| Volume Distribution (VD): | 1.226 | Fu: | 84.66% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.29 | CYP1A2-substrate: | 0.661 |
| CYP2C19-inhibitor: | 0.126 | CYP2C19-substrate: | 0.856 |
| CYP2C9-inhibitor: | 0.02 | CYP2C9-substrate: | 0.795 |
| CYP2D6-inhibitor: | 0.012 | CYP2D6-substrate: | 0.827 |
| CYP3A4-inhibitor: | 0.012 | CYP3A4-substrate: | 0.238 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 6.926 | Half-life (T1/2): | 0.741 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.009 | Human Hepatotoxicity (H-HT): | 0.147 |
| Drug-inuced Liver Injury (DILI): | 0.049 | AMES Toxicity: | 0.047 |
| Rat Oral Acute Toxicity: | 0.011 | Maximum Recommended Daily Dose: | 0.03 |
| Skin Sensitization: | 0.183 | Carcinogencity: | 0.031 |
| Eye Corrosion: | 0.012 | Eye Irritation: | 0.462 |
| Respiratory Toxicity: | 0.365 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
| ENC000289 | 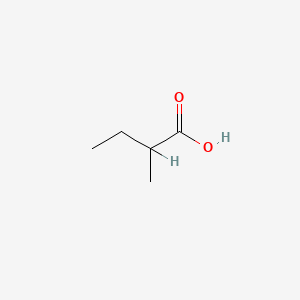 |
0.296 | D0ZK8H |  |
0.267 | ||
| ENC001474 | 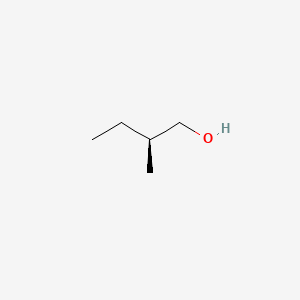 |
0.269 | D0M1PQ |  |
0.222 | ||
| ENC000307 | 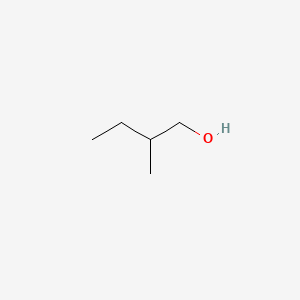 |
0.269 | D0W0MF |  |
0.176 | ||
| ENC000182 | 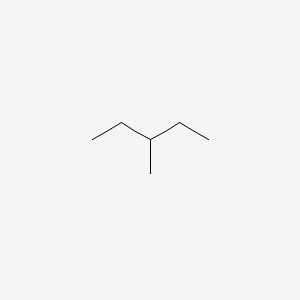 |
0.269 | D0P6UB |  |
0.167 | ||
| ENC000619 |  |
0.267 | D03DVJ |  |
0.167 | ||
| ENC000225 | 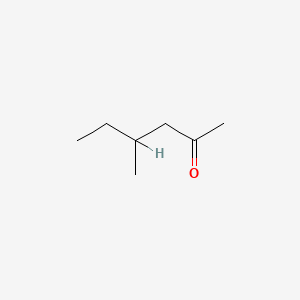 |
0.267 | D0A4JK |  |
0.163 | ||
| ENC000001 | 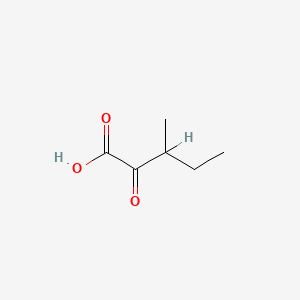 |
0.250 | D0A8CJ | 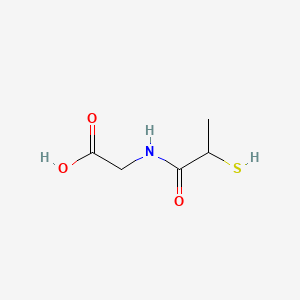 |
0.162 | ||
| ENC000141 |  |
0.250 | D0Y3KG |  |
0.158 | ||
| ENC000771 | 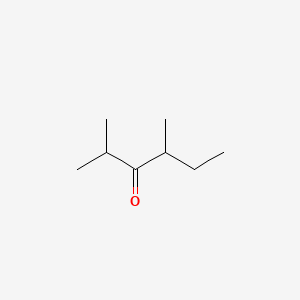 |
0.250 | D02KBD | 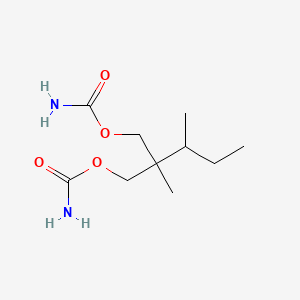 |
0.157 | ||
| ENC000874 | 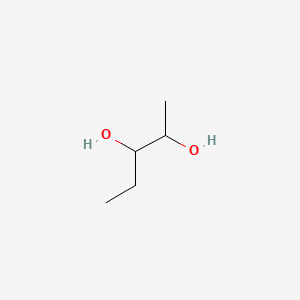 |
0.250 | D07ZTO | 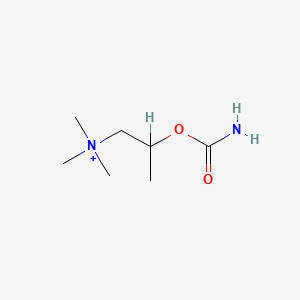 |
0.154 | ||