Drug General Information
| Drug Name |
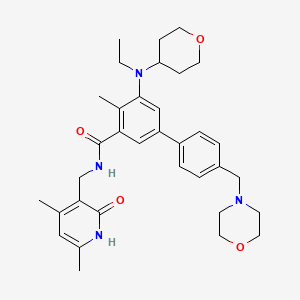
Saroglitazar
|
||
| Synonyms |
Lipaglyn; 495399-09-2; UNII-E0YMX3S4JD; E0YMX3S4JD; Saroglitazar magnesium; (S)-2-Ethoxy-3-(4-(2-(2-methyl-5-(4-(methylthio)phenyl)-1H-pyrrol-1-yl)ethoxy)phenyl)propanoic acid; Saroglitazar [INN]; saroglitazarum; SCHEMBL16346340; DTXSID10197819; MRWFZSLZNUJVQW-DEOSSOPVSA-N; CHEBI:134708; AKOS027337115; CS-6149; DB13115; HY-19937; (2S)-2-ethoxy-3-[4-(2-{2-methyl-5-[4-(methylthio)phenyl]-1H-pyrrol-1-yl}ethoxy)phenyl]propanoic acid; (S)-alpha-ethoxy-4-[2-[-methyl-5-[4-(methylthio) phenyl]-1H-pyrrol-1-yl]ethox
|
||
| Drug Type |
NA
|
||
| Indications |
Dyslipidemia [ICD-11: 5C80-5C81]
|
Approved
|
|
| Company |
Zydus Cadila Gujarat, India
|
||
| Summary |
Saroglitazar has been investigated for the treatment of Fatty Liver.
|
||
Drug Chemical Infomation and External Links
|
Formula |
C25H29NO4S
|
| Canonical SMILES |
CCOC(CC1=CC=C(C=C1)OCCN2C(=CC=C2C3=CC=C(C=C3)SC)C)C(=O)O
|
|
| InChI |
1S/C25H29NO4S/c1-4-29-24(25(27)28)17-19-6-10-21(11-7-19)30-16-15-26-18(2)5-14-23(26)20-8-12-22(31-3)13-9-20/h5-14,24H,4,15-17H2,1-3H3,(H,27,28)/t24-/m0/s1
|
|
| InChIKey |
MRWFZSLZNUJVQW-DEOSSOPVSA-N
|
|
| CAS Number | CAS 495399-09-2 | |
| TTD ID | D0E8LA | |
| DrugBank ID | DB13115 | |
| PubChem Compound ID | 60151560 | |
| PubChem Substance ID |
NA
|
|
| ChEBI ID | CHEBI:134708 | |
| ADReCS Drug ID | NA |
Drug-Food (Herb) Interactions
- Drug
- Foods
- Herbs
- Positive
- Negative
- Possible
- Harmful
- No Effect
| ID | Name | Dose | Type | Drug Brand Name | Drug Dose | Drug Dosage Form | Experimental Species | Individuals Number | Test Sample | Ingredient | Effect | Relationship Classification | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ID | Name | Dose | Type | Drug Brand Name | Drug Dose | Drug Dosage Form | Experimental Species | Individuals Number | Test Sample | Ingredient | Effect | Relationship classification |