Drug General Information
| Drug Name |
Valsartan
|
||
| Synonyms |
valsartan; 137862-53-4; Diovan; Tareg; Provas; L-Valsartan; CGP 48933; Exforge; CGP-48933; (S)-2-(N-((2'-(1H-Tetrazol-5-yl)-[1,1'-biphenyl]-4-yl)methyl)pentanamido)-3-methylbutanoic acid; UNII-80M03YXJ7I; N-(p-(o-1H-Tetrazol-5-ylphenyl)benzyl)-N-valeryl-L-valine; CHEMBL1069; 80M03YXJ7I; CHEBI:9927; C24H29N5O3; (2S)-3-methyl-2-[pentanoyl-[[4-[2-(2H-tetrazol-5-yl)phenyl]phenyl]methyl]amino]butanoic acid; N-(1-oxopentyl)-N-[[2'-(1H-tetrazol-5-yl)[1,1'-biphenyl]-4-yl]methyl]-L-valine; 137863-60-6; AK-58790; Kalpress; Miten; Nisis; Diovan; Vals; Valsarran; Walsartan; Aventis brand of valsartan; CEPA brand of valsartan; Esteve brand of valsartan; Lacer brand of valsartan; Novartis brand of valsartan; Sanol brand of valsartan; Schwarz brand of valsartan; Diovan (TN); Diovan, Valsartan; Valsartan [USAN:INN]; Valtan (TN); Valzaar (TN); Valsartan (JAN/USAN/INN); N-valeryl-N-((2'-(1H-tetrazol-5-yl)biphenyl-4-yl)methyl)valine; N-pentanoyl-N-{[2'-(1H-tetrazol-5-yl)biphenyl-4-yl]methyl}-L-valine; N-pentanoyl-N-{[2'-(1H-tetrazol-5-yl)[1,1'-biphenyl]-4-yl]methyl}-L-valine; L-Valine, N-(1-oxopentyl)-N-[[2'-(1H-tetrazol-5-yl)[1,1'-biphenyl]-4-yl]methyl]-(9CI); (S)-N-valeryl-N-{[2'-(1H-tetrazol-5-yl)biphenyl-4-yl]-methyl}-valine; (s)-2-(n-((2'-(1h-tetrazol-5-yl)biphenyl-4-yl)methyl)pentanamido)-3-methylbutanoic acid; [3H]valsartan
|
||
| Drug Type |
Small molecular drug
|
||
| Indications |
Hypertension [ICD-11: BA00-BA04]
|
Approved
|
|
| Company |
Norvatis Phamaceuticals Corporation
|
||
| Summary |
Valsartan is an angiotensin-receptor blocker used to manage hypertension alone or in combination with other antihypertensive agents and to manage heart failure in patients who are intolerant to ACE inhibitors.
|
||
| Target |
Angiotensin II receptor type-1 (AGTR1)
Mechanism of Action: Antagonist
|
T74456 | |
Drug Chemical Infomation and External Links
|
Formula |
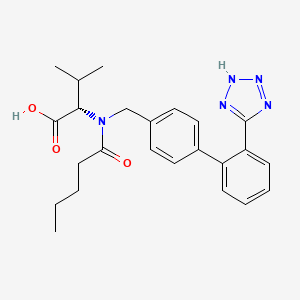
C24H29N5O3
|
| Canonical SMILES |
CCCCC(=O)N(CC1=CC=C(C=C1)C2=CC=CC=C2C3=NNN=N3)C(C(C)C)C(=O)O
|
|
| InChI |
1S/C24H29N5O3/c1-4-5-10-21(30)29(22(16(2)3)24(31)32)15-17-11-13-18(14-12-17)19-8-6-7-9-20(19)23-25-27-28-26-23/h6-9,11-14,16,22H,4-5,10,15H2,1-3H3,(H,31,32)(H,25,26,27,28)/t22-/m0/s1
|
|
| InChIKey |
ACWBQPMHZXGDFX-QFIPXVFZSA-N
|
|
| CAS Number | CAS 137862-53-4 | |
| TTD ID | D06UDG | |
| DrugBank ID | DB00177 | |
| PubChem Compound ID | 60846 | |
| PubChem Substance ID |
7847466
,
11066615
,
11364700
,
11367262
,
11369824
,
11373100
,
11374217
,
11377988
,
11484996
,
11488936
,
11491646
,
11492564
,
11495610
,
14856825
,
14881173
,
26612829
,
26719825
,
43118184
,
46386599
,
46509000
,
46530915
,
47499543
,
48393917
,
49681716
,
49830875
,
50062253
,
50467452
,
53787275
,
57314146
,
81093312
,
85788951
,
90452226
,
92124805
,
92308052
,
92308462
,
92711441
,
93166503
,
99228303
,
103292815
,
103979540
,
104253413
,
104321799
,
117541337
,
117664453
,
119526522
,
123055291
,
124659015
,
124757534
,
124800101
,
125164338
|
|
| ChEBI ID | CHEBI:9927 | |
| ADReCS Drug ID | BADD_D02331 |
Drug-Food (Herb) Interactions
- Drug
- Foods
- Herbs
- Positive
- Negative
- Possible
- Harmful
- No Effect
| ID | Name | Dose | Type | Drug Brand Name | Drug Dose | Drug Dosage Form | Experimental Species | Individuals Number | Test Sample | Ingredient | Effect | Relationship Classification | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ID | Name | Dose | Type | Drug Brand Name | Drug Dose | Drug Dosage Form | Experimental Species | Individuals Number | Test Sample | Ingredient | Effect | Relationship classification |